Self-assembly of cell division machinery in space and time in E. coli
The dream of every bacterial cell is to become two cells (Francois Jacob). How this dream comes about? Most bacteria divide by building cell wall in the center of the cell. The building process needs to go hand-in-hand with chromosome replication to guarantee that each daughter cell receives full complement of genetic material. If this conditon is not met then the cell dies.
 |
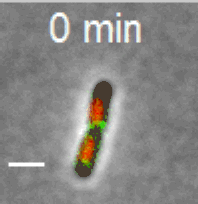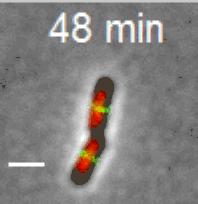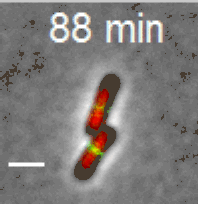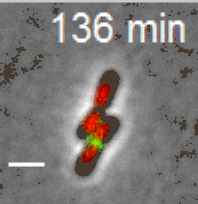
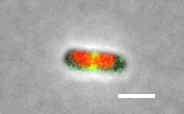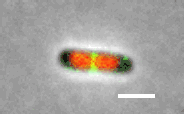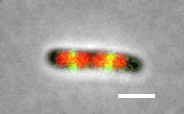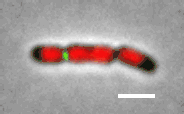
Cell division in E. coli. Red color labels chromsomes and green cell division apparatus. Scale bar is 2 um. |
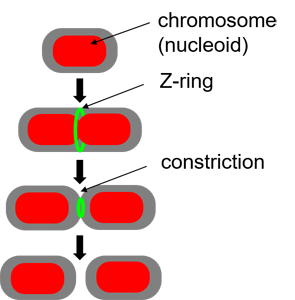
The building starts with the assambly of FtsZ (proto)filaments in the center of the cell to a ring-like structure, the Z-ring (see Figure above). A scaffold fromed of FtsZ protofilements recruits more than 30 different proteins that carry out septal cell wall synthesis and gurantee that the daughter chromsomes become correctly partitioned to new cells. The aim of our research is to understand the initial self-assembly process of the FtsZ filaments at the center of the cell and how different cellular regulators affect this assembly process. FtsZ filaments are unlike organic polymers synthesized in chemistry lab. They consists of protein monomers that are constantly leaving and joining the filament; leaving mostly from one end of the filament and joining from another. This treadmilling type of shuffling requires steady energy input. This highly dynamic assembly appears to be essential in controlling where and when the filaments form but at the same time the rapid changes in the structure make studying this system difficult and higher order stucture of these filament is not yet known.
Positioning of the Z-ring
Several molecular systems have been identified in E. coli that are involved in positioning its cell division apparatus. The most well-known of them is the Min system that oscillates back and forth in the cell. In addition to the Min system, a so-called nucleoid occlusion factor SlmA has been identified that prevents the filament assembly at certain locations of the chromosome (all but at the replication Terminus region). Our research have brought out yet additional protein system invovlved in positioning the cell divison apparatus. We refer to this system as the Ter linkage. While the Min system and SlmA regulate the assembly of FtsZ protofilaments via inhibition (negative regulation), the Ter linkage promotes the assembly of cell division proteins at the replication terminus region (Movie Below Left). It thus implements a positive regulatory mechanism.
For a cell lacking the Min system and nucleoid occlusion factor, the Z-ring jumps form from cell middle to its 1/4 positions after duplication of the replication terminus region. |
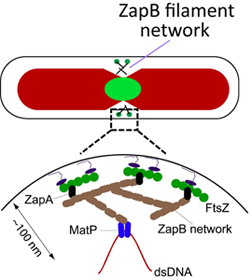
Possible arrangement of the Ter linkage proteins |
The key elements of the Ter linkage are MatP, ZapA and ZapB proteins that together connect chromosomal DNA in replication terminus region to FtsZ protofilaments. Although the exact structure of this centrosome-like assembly is not yet clear it may look like as shown on the top right Figure. Our interest is to further understand the structure of the Ter linkage and how it forms during cell cycle.
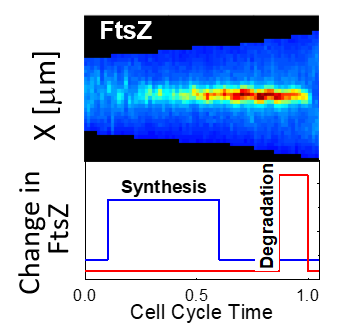
Timing of the Z-ring
What times the coordination of FtsZ assembly with cell division is not currently known. We have recently shown that there is a cell-cycle control of FtsZ levels and upregulation of these levels correllates with the fromation of the Z-ring. Important part of cell cycle regulation of FtsZ levels is its rapid degradation by ClpPX protease at the end of the cell cycle. This degradation removes larger number of proteins at slow growth conditions. However, our data indicates that upregulation of FtsZ levels is just of one factor that is needed for Z-ring formation. The search is on for additional factors that are needed to trigger this essential cellular process.