Development microfluidic devices for bacterial studies
Microfluidics has a significant potential to advance cellular studies via more precise control of cellular environment, automation and high throughput. Our goal is to develop microfluidic platforms that enable delivery of variety of chemical and physical stimuli to individual bacterial cells and to observe cellular response as it unfolds in real time using optical microscopy. The chips that we develop can be grouped as the ones that deliver mechanical stimuli (squeezing chips) and the ones that deliver chemical ones (mother machine type devices; see below).
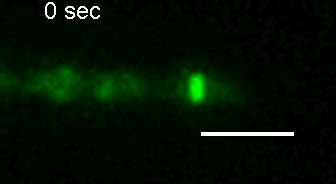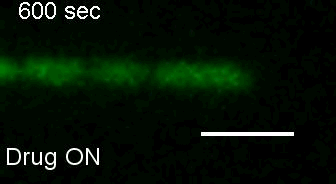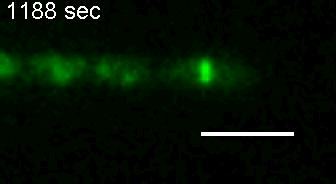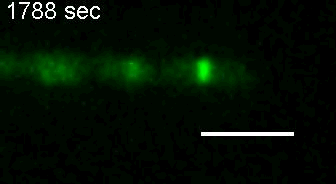
E. coli Z-ring during pulsing with a drug. |
Squeezing chips
In past years we have developed both active and passive chips to study bacterial cellular response to mechanical perturbations. In passive chips the cells are confined simply by the channel walls. Under resulting uniaxial mechanical stress E. coli transform from regular rod-shaped cells to variety of different morphologies.
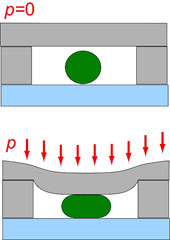
In active chips we use pneumatic microscale valves to exert uniaxial stresses to cells (Figure below). Despite cell wall, E. coli cells are flexible and allow considerable stretching of the cell wall (strains up to 20%).
A cross-sectional view of a pressure-actuated microvalve which is used to apply uniaxial stress to a bacterial cell (green cricle). |
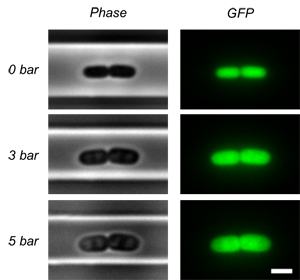
Phase contrast (left) and fluoresence (right) images of an E. coli cell under the valve at different closing pressures of the valve. |
Mothermachine platform
We have been also working on further improvements on mother machine platform. In particular we have been using agarose as the material for the channels. While agarose is not easy to handle, it allows much better optical quality of the chips becuase the refractive index of channel material is very close to refractive index of cell medium.
E. coli cells in agarose based mothermachine chips |